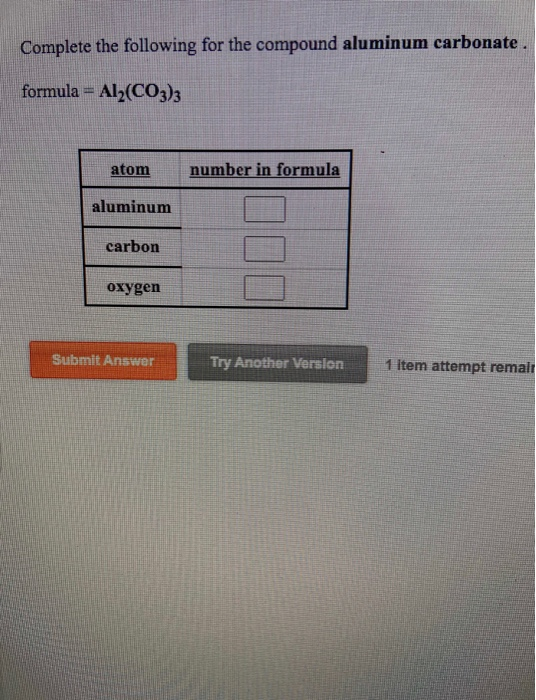
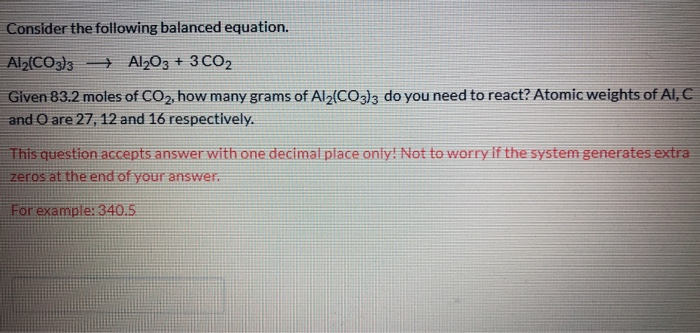
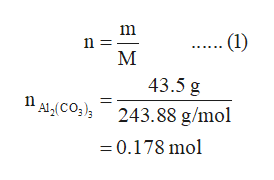SOLVED: 1point What is the molecular formula for aluminum carbonate? Als (CO:)2 AICO: Alz (CO3)3 AlzC309

Complete the following table below by writing the correct formula for the compound formed between each combination of cation and anion. Then write the correct chemical name for each. CO3 2? PO4